Addiction is a multifaceted topic that has been studied from multiple perspectives and talked about in a variety of ways. Most of the discussion has focused on the compulsive ingestion of a chemical substance, whether legal and socially accepted, such as caffeine, alcohol, and various prescription drugs; or illegal and considered socially unacceptable, like heroin or methamphetamines. In recent years, the definition of addiction has moved beyond chemical substances to include other types of compulsive behavior or activities, such as gambling or the viewing of pornographic images.
It is quite likely that the characteristics of many of these behavioral compulsions resemble those associated with chemical addictions. Behaviors that have been linked to addictions in the psychological literature include gambling, 1 Internet use,4 pornography,6 though the evidence is mixed for at least some of these. 7
This article will provide an overview of the biological processes that accompany chemical addictions and review the evidence that these processes also accompany behavioral addictions. I will then suggest reasons why these behaviors are so resistant to change, and discuss the role of choice in the ways in which these addictions manifest themselves. I will finally suggest how neurological processes and faith might intersect as we consider the phenomenon of addiction. But first, a working definition of addiction.
What Is an Addiction?
Traditionally, addiction has been associated with the ingestion of a chemical substance. For example, Hyman defined addiction as “compulsive drug use despite negative consequences,” 8 which causes individuals to increasingly narrow their attention to the task of obtaining and ingesting the drug, and/or recovering from the use of said drugs, despite competing family needs, failing health, and potential and actual threats to their freedom. However, Marks 9 makes room for a much broader and more encompassing definition, allowing for the inclusion of behavioral addictions—any repetitive routine with a frequency or intensity that leads to restricted behavior. Alavi et al. 10 recognize that the definition of addiction can be controversial, yet they also view dependence on a substance or activity as the central feature.
The World Health Organization (WHO)-sponsored International Classification of Diseases (ICD-10) has shown a preference for the term dependence rather than addiction and defines a dependence syndrome as “a cluster of physiological, behavioural, and cognitive phenomena in which the use of a substance or a class of substances takes on a much higher priority for a given individual than other behaviours that once had greater value.” 11 The statement goes on to list criteria for diagnosis: (a) strong desire or compulsion, (b) physiological withdrawal when use has ceased, (c) evidence of tolerance, (d) progressive neglect of alternative pleasures or interests, and (e) persistent use even in the face of harm. The recently released fifth edition of the American Psychiatric Association’s Diagnostic and Statistical Manual of Mental Disorders (DSM-5), includes the category of “Substance-related and Addictive Disorders” with similar criteria as the ICD-10 and does not use the term dependence at all. However, in this latest edition, gambling is listed for the first time as being addictive, while Internet games, though not listed as addictive, are flagged for further study. 12 So it seems fair to say that a consensus is developing to include not only chemical substances, but also certain behaviors within any working definition of addiction.
It should be noted that not all compulsive behaviors are addictions. As Marks 13 indicates, many of our daily survival behaviors like eating, drinking, and even sex are behaviors in which the desire to engage in them increases over time, and this desire is reduced once the act is completed, then returns after a few hours or days. Yet we do not call these behaviors addictions unless they become dysfunctional and go beyond the functional need to sustain life or well-being. Nor do we include non-purposeful dysfunctional behaviors, such as Tourette’s syndrome or other compulsive disorders. 14 Thus, by definition, addictions include only behaviors that are still under voluntary, though diminished, control.
Neurological Processes in Addiction
First, some background. All behavior, as far as we know, involves the transmission of information through nerve cells and from one nerve cell to another. The nerve cells are often found in bundles called nuclei, and form pathways or nerves made of bundles of axons. Information transmission occurs in two ways. First, transmission within nerve pathways is by electrical impulses along individual axons. Next, transmission from one cell to another involves chemical substances called neurotransmitters that carry these messages by activating protein structures in the nerve cell called receptors. Common neurotransmitters include dopamine, serotonin, gamma amino butyric acid (GABA), opioid peptides, and glutamate.15
Whenever we do something that makes us feel good or satisfies some innate desire, whether to taste a piece of cheesecake or receive a sum of money, the resultant pleasure comes from the activation of reward systems in the brain. These systems involve areas in the cortex (orbito-frontal and insula) as well as subcortical areas such as the nucleus accumbens and the amygdala, together called the mesocorticilimbic (or hedonic) circuit (see Figure 1). These structures include circuits for a variety of neurotransmitters such as endogenous opioid peptides and dopamine. 16 This occurs when the behavior is normal and functional, as well as when the behavior is dysfunctional.
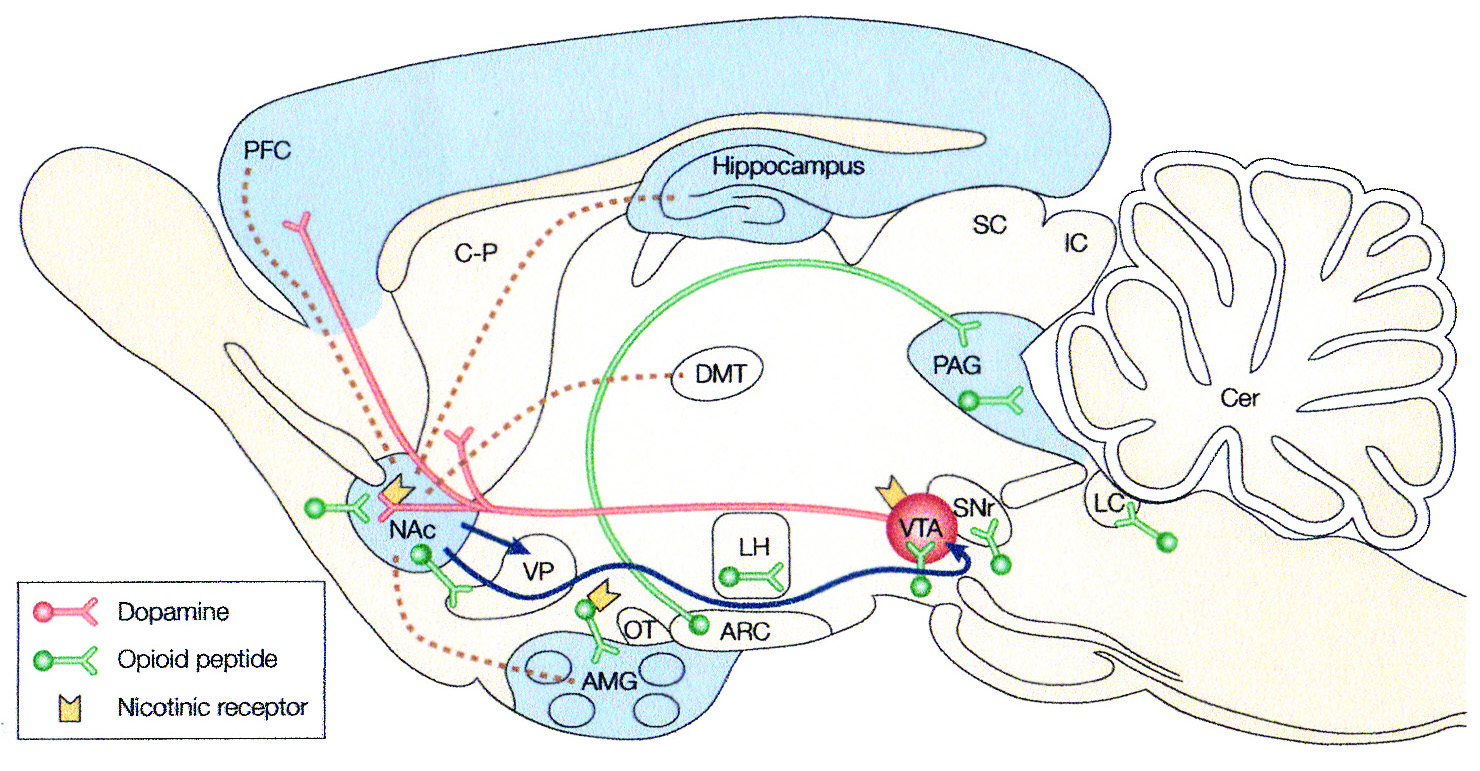
Figure 1. Key Neural Circuits of Addiction
Dotted lines indicate limbic afferents to the nucleus accumbens (NAc). Blue lines represent efferents from the NAc thought to be involved in drug reward. Red lines indicate projections of the mesolimbic dopamine sys-tem thought to be a critical substrate for drug reward. Dopamine neurons originate in the ventral tegmental area (VTA) and project to the NAc and other limbic structures, including the olfactory tubercle (OT), ventral do-mains of the caudate-putamen (C-P), the amygdala (AMG), and the prefrontal cortex (PFC). Green indicates opioid-peptide-containing neurons, which are involved in opiate, ethanol, and possibly nicotine reward. These opioid peptide systems include the local enkephalin circuits (short segments) and the hypothalmic midbrain ß- endorphin circuit (long segment). Blue shading indicates the approximate distribution of GABAA (y-aminobutyric acid) receptor complexes that might contribute to ethanol reward. Yellow solid structures indicate nicotinic acetylcholine receptors hypothesized to be located on dopamine- and opoid-peptide-containing neurons (ARC, arcuate nucleus; Cer, cerebellum; DMT, dorsomedial thalamus; lC, inferior colliculus; LC, locus coeruleus; LH, lateral hypothalamus; PAG, periaqueductal grey; SC, superior colliculus; SNr, substantia nigra pars reticulata; VP, ventral pallidum).
Reprinted by permission from Macmillan Publishers Ltd.: Eric J. Nestler, “Molecular Basis of Long-term Plas-ticity Underlying Addiction,” Nature Reviews Neuroscience 2 (February 2001):121.
A review of research on pleasure by Berridge and Kringelbach 17 suggests that reward involves at least three related aspects: (1) the feeling of pleasure or liking, (2) the motivation (or wanting) to obtain what is liked, and (3) the learning, or behavior change, that results from obtaining a reward. Humans experience different subjective feelings of liking, depending on the object. Thus, the pleasurable feelings associated with a sexual orgasm, an opoid drug, or a beautiful piece of music may seem different. Yet, according to Berridge and Kringelbach, these all seem to arise from activity in the mesocorticolimbic circuit. 18
According to Kalivas and Volkow,19 addiction to drugs appears to progress through three stages. In the first stage, the rewarding effects of the drug are accompanied by the release of dopamine into the nucleus accumbens, which seems to be more related to the “wanting” or motivational aspect of the reward system than the “liking” aspect. 20 This dopamine release is also associated with short-term changes in gene expression in the neuron itself. The gene changes are temporary because the protein produced is unstable and persists for only a few hours. 21 So the pleasant feeling caused by the drug endures briefly, and addiction has not yet begun.
Christians have the explicit promise that a “way of escape” from every temptation is provided if we trust in God who “is faithful.” One can think of few temptations more powerful than those represented by behavioral addictions. this promise, and others like it throughout Scripture, provide the assurance that reliance on God and trust in his faithfulness can be a powerful resource for overcoming the most enslaving addiction.
Continued drug use takes the individual to the second stage, transitional to addiction. In this stage, repeated activation of the dopamine receptor influences the accumulation of a more stable protein (called ΔFosB). The increased presence of ΔFosB protein appears to be related to more permanent structural changes in the cells of the nucleus accumbens, the hippocampus, and other structures in the reward system. Among these changes is an expanded branching in the dendritic tree and proliferation of dendritic spines, producing increased rewarding effects of the drug (see Figure 2).
The final stage (or end-stage) of drug addiction involves increased vulnerability to relapse caused by further changes to the nervous system’s cellular structure. Paradoxically, these changes become greater with increased periods of withdrawal. These changes in the cell “convert vulnerability to relapse from temporary and reversible into permanent features of addiction.” 22 As a result, the addicted person finds it harder and harder to quit with each failed attempt to do so. This is described by Olsen 23 as the hijacking of the reward system, leading to the dysfunctional effects associated with these drugs.
The evidence now suggests that this process applies as well with a variety of rewarding behaviors under certain conditions. Twenty-five years ago, Marks 24 argued that, based on the ICD definition of a dependence syndrome, “the urge of behavioural addicts to engage in their behavioural routine, and the discomfort ensuing if prevented from completing it, respectively resemble the craving and the withdrawal symptoms of substance abusers.” 25 He further noted that some withdrawal symptoms are the same across the two categories, while others are substance-specific. More apropos to this discussion, he argued that some brain mechanisms are common to the establishment and maintenance of all addictions, whether behavioral or substance-related. Grant et al. 26 also claim that evidence increasingly indicates that behavioral addiction and substance use disorders share common features both in terms of cognition and neurotransmitter systems.
If addiction is caused by the extraordinary effect of dopamine on the reward system by drugs, do ordinary behaviors produce those effects as well? Clearly, not all rewarding behavior produces addictive effects—only the ones that produce artificially high rewards transcend those of ordinary day-to-day events. Only activities such as gambling, gaming, and pornography produce such high dopamine secretions as to result in addiction. It also appears that some individuals are genetically programmed for an addictive response. This is further supported by the finding that behavioral and substance addictions are often found in the same individual 27 and in people who are genetically related. 28
Addiction and Choice
Describing addiction in chemical and mechanistic terms, and observing the consequent resistance to change associated with addictions, would naturally lead one to ask whether, in fact, addicted persons have any choice in the matter. Or might personal choice in some way modify those neurological processes? There are two aspects to the issue of choice in addictions: (1) the choice to engage in a particular behavior that eventually becomes addictive; and (2) the choice to continue to engage in a behavior after addiction has become established.
With regard to the initial choice to engage in behavior that is potentially addictive, one can argue that individual freedom is unimpaired prior to addiction; therefore, he or she bears full responsibility whether to act or not to act. Yet even at this stage, genetic factors seem to intervene, influencing voluntary choice. A Minnesota study of twins reared apart indicated a strong correlation in religious beliefs among identical twins and much weaker or nonexistent correlations among fraternal twins. 29 Similarly, a Dutch study found that sensation-seeking behaviors were also tied to hereditary factors. 30 As Gene Heyman has argued, “religious beliefs are voluntary; genes affect religious beliefs; [therefore] genes affect voluntary behavior.” 31
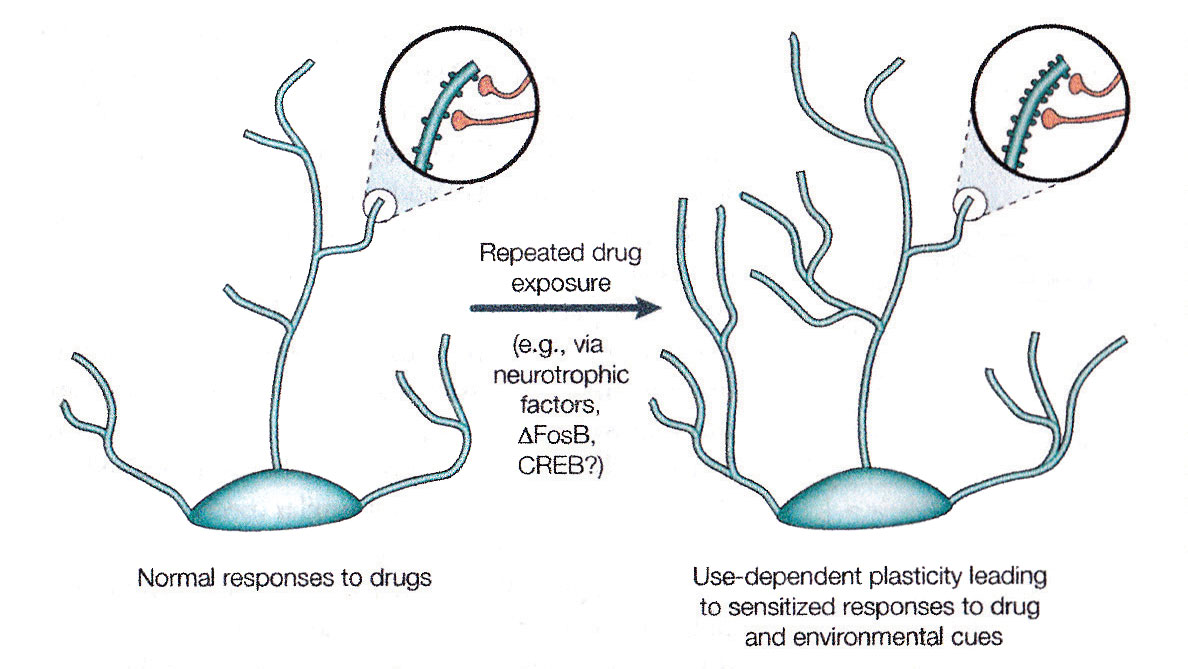
Figure 2. Regulation of Dendritic Structure by Drugs of Abuse
The figure shows the expansion of a dentritic tree after chronic exposure to a drug of abuse, as has been observed in the nucleus accumbens and in the prefrontal cortex. The areas of magnification show an increase in dendritic spines, which is postulated to occur in conjunction with activated nerve terminals. Such alterations in dentritic structure, which are similar to those observed in other exam-ples of synaptic plasticity such as long-term potentiation, could mediate long-lived sensitized re-sponses to drugs of abuse or environmental cues.
Reprinted by permission from Macmillan Publishers Ltd: Eric J. Nestler, “Molecular Basis of Long-term Plasticity Un-derlying Addiction,” Nature Reviews Neuroscience 2 (February 2001):126.
However, genetic influence does not mean absence of choice. A closer examination of the Minnesota study shows that even among the identical twins, the variance explained is less than 50 percent, meaning that factors other than genes, including choice, explain more than half of the variance. Therefore, although genes may make certain behaviors more likely to occur, there is much room for individual choice to determine whether to engage in potentially addictive behaviors.
The role of choice after an addiction has set in is even more interesting, since addiction is often defined in terms of the compulsion to engage in a particular behavior. 32 As discussed earlier, addiction involves changes in the brain that tend to reduce impulse control and increase the craving (or wanting) for the reward that comes with a particular behavior. This increased craving may be accompanied by withdrawal effects that negatively reinforce the behavior. Yet, Heyman insists that “addiction is a disorder of choice.” 33 He argues that the data show that most persons who meet the criteria for addiction are able to quit by age 30; most quit without professional help; and the reasons for quitting include legal concerns, economic concerns, and the desire for respect, especially from family members. He concludes “the correlates of quitting are the correlates of choice not compulsion.” 34
Heyman’s argument, though persuasive, contains the defect of being unnecessarily binary. Addiction involves neither total compulsion nor unfettered choice. Rather, because of the changes to the reward systems described earlier, addiction involves a disruption of the mechanisms of choice, with factors such as withdrawal making choice more difficult, increasing the likelihood of self-destructive behavior, although this is not an inevitable result. Yet, just as the brain is changed by addictive behavior, changes to the brain also occur by the exercise of voluntary behavior. Thus, brain plasticity also ensures the opportunity for recovery.
The Role of Faith
Heyman reports that addicts often say that they quit drugs because “they wanted to be a better parent, make their own parents proud of them, and not further embarrass their families.” 35 In each of these cases, the addict made a choice. Since these motivations are likely to be enhanced by religious faith, it may be expected that religious faith could be a valuable pathway to ending addiction. Accordingly, Hansen 36 has reported that faith-based treatments have been helpful in the treatment of addiction in Puerto Rico. Researchers have shown the efficacy of prayer and meditation on self-regulation and self-control. 37 A number of studies have shown the effects of meditation 39 on brain plasticity. However, McCullough and Willoughby noted that a range of activities are encompassed by the terms prayer and meditation, and that not all such activities are likely to have the same salutary effects. 40 One might add that none of these activities is specifically Christian, and some can be readily identified with religious practices connected to other religious traditions. A degree of caution is therefore warranted in interpreting the extant research.
However, the evidence provides reason for hope that, although addiction involves definite and profound changes to brain structure and function, the human brain is created with the capacity for change, and that change can come through the exercise of choice, aided by religious practices such as prayer, meditation, and dependence on divine power.
Christians have the explicit promise that a “way of escape” from every temptation is provided if we trust in God who “is faithful.” 41 One can think of few temptations more powerful than those represented by behavioral addictions. This promise, and others like it throughout Scripture, 42 provide the assurance that reliance on God and trust in His faithfulness can be a powerful resource for overcoming the most enslaving addiction.
This article has been peer reviewed.
Recommended citation:
Austin C. Archer, “Biological Aspects of Addictive Behavior,” The Journal of Adventist Education 78:4 (April–May 2016): 8-12. Available at https://www.journalofadventisteducation.org/en/2016.4.3.
NOTES AND REFERENCES
- R. I. Brown, “Gambling Addictions, Arousal, and an Affective/Decision-making Explanation of Behavioral Reversions or Relapses,” The International Journal of the Addictions 22:11 (May 1987):1053-1067. doi: 10.3109/10826088709027469; Nady El-Guebaly et al., “Compulsive Features in Behavioral Addictions: The Case of Pathological Gambling,” Addiction 107:10 (October 2012):1726-1734. doi: 10. 1016/j.biotechadv.2011.08.021; Luke Clark and Eve H. Limbrick-Oldfield, “Disordered Gambling: A Behavioral Addiction,” Current Opinion in Neurobiology 23:4 (August 2013):655-659. doi: 10.1016/j.conb.2013.01.004.
- Kathryn Yung et al., “Internet Addiction Disorder and Problematic Use of Google GlassTM in Patient Treated at a Residential Substance Abuse Treatment Program,” Addictive Behaviors 41 (September 2014):58-60. doi: 10.1016/j. addbeh.2014.09.024.
- Derek D. Reed, “Ultra-violet Indoor Tanning Addiction: A Reinforcer Pathology Interpretation,” Addictive Behaviors 41 (February 2015):247-251. doi: 10.1016/j.ad dbeh.2014.10.026.
- Sergey Krivoschekov and O. N. Lushnikov, “Psychophysiology of Sports Addictions (Exercise Addiction),” Human Physiology 37:4 (July-August 2011):509-513. doi: 10.1134/S0362119711030030.
- Clark Watts and Donald Hilton, “Pornography Addiction: A Neuroscience Perspective,” Surgical Neurology International 2:1 (February 2011):19. doi: 10. 4103/2152-7806.76977.
- Kent C. Berridge and Morten L. Kringelbach, “Neuroscience of Affect: Brain Mechanisms of Pleasure and Displeasure,” Current Opinion in Neurobiology 23:3 (June 2013):294-303./j.conb.2013.01.017. See also Ann E. Kelley and Kent C. Berridge, “The Neuroscience of Natural Rewards: Relevance to Addictive Drugs,” The Journal of Neuroscience 22:9 (May 2002):3306-3311.
- Kristen Weir, “Is Pornography Addictive?” Monitor on Psychology (April 2014): http://www.apa.org/monitor/2014/04/pornography.aspx. Accessed July 16, 2015.
- Steven E. Hyman, “Addiction: A Disease of Learning and Memory,” The American Journal of Psychiatry 162:8 (August 2005):1414-1422.
- Isaac Marks, “Behavioural (Non-chemical) Addictions,” British Journal of Addictions 85:11 (October 1990a):1389-1394.
- Seyyed Salman Alavi et al,, “Behavioral Addiction Versus Substance Addiction: Correspondence of Psychiatric and Psychological Views,” International Journal of Preventive Medicine 3:4 (April 2012):290-294.
- World Health Organization, Management of Substance Abuse (2015): http://www.who.int/substance_abuse/terminology/definition1/en/. Accessed June 7, 2015.
- American Psychiatric Association, Highlights of Changes From DSM-IV-TR to DSM 5 (2013): http://www.dsm5.org/Documents/changes%20from%20dsm-iv-tr%20to%20dsm-5.pdf. Accessed July 15, 2015.
- Marks, “Behavioural (Non-chemical) Addictions,” op. cit.
- __________, “Reply to Comments on ‘Behavioural (Non-chemical) Addictions,’” British Journal of Addictions 85:11 (November 1990b):1429-1431.
- Neil V. Watson and S. Marc Breedlove, The Mind’s Machine: Foundations of Brain and Behavior , 2nd ed. (Sunderland, Mass.: Sinauer Associates, Inc., 2016).
- K. S. LaForge, V. Yuferov, and M. J. Kreek, “Opioid Receptor and Peptide Gene Polymorphisms: Potential Implications for Addictions,” European Journal of Pharmacology 410:2-3 (2000):249-268. doi: 10.1016/S0014-2999(00)00819-0.
- Berridge and Kringelbach, “Neuroscience of Affect,” op cit. See also A. E. Kelley and K. C. Berridge, “The Neuroscience of Natural Rewards: Relevance to Addictive Drugs,” The Journal of Neuroscience 22:9 (May 2002):3306-3311.
- Ibid.
- Peter W. Kalivas and Nora D. Volkow, “The Neural Basis of Addiction: A Pathology of Motivation and Choice,” American Journal of Psychiatry 162:8 (August 2005):1403-1413. doi: 10.1176/appi.ajp.162.8.1403.
- Berridge and Kringelbach, “Neuroscience of Affect,” op. cit.
- Eric J. Nestler, “Molecular Basis of Long-term Plasticity Underlying Addiction,” Nature Reviews Neuroscience 2 (February 2001):119-128.
- Kalivas and Volkow, “The Neural Basis of Addiction,” op. cit., p. 1408.
- Christopher M. Olsen, “Natural Rewards, Neuroplasticity, and Non-Drug Addictions,” Neuropharmacology 61:7 (December 2011):1109–1122. doi: 10.1016/j.neu ropharm.2011.03.010.
- Marks, “Behavioural (Non-chemical) Addictions,” op. cit.
- Ibid., p. 1391.
- J. E. Grant et al., “Introduction to Behavioral Addictions,” American Journal of Drug and Alcohol Abuse 36:5 (September 2010):233-241.
- Ibid.
- Donald W. Black et al., “Family History and Psychiatric Comorbidity in Persons With Compulsive Buying: Preliminary Findings,” American Journal of Psychiatry 155:7 (July 1998):960-963; Judson A. Brewer and Marc N. Potenza, “The Neurobiology and Genetics of Impulse Control Disorders: Relationships to Drug Addictions,” Biochemical Pharmacology 75:1 (January 2008):63-75. doi: 10.1016/ j.bcp.2007.06.043.
- Niels G. Waller et al., “Genetic and Environmental Influences on Religious Interests, Attitudes, and Values: A Study of Twins Reared Apart and Together,” Psychological Science 1:2 (1990):138-142.
- R. D. Stoel, E. J. De Geus, and D. I. Boomsma, “Genetic Analysis of Sensation Seeking With an Extended Twin Design,” Behavior Genetics 36:2 (March 2006): 229-237.
- G. M. Heyman, Addiction: A Disorder of Choice (Cambridge: Harvard University Press, 2009), p. 173.
- Hyman, “Addiction: A Disease of Learning and Memory,” op. cit.
- Heyman, Addiction: A Disorder of Choice, op. cit., p. 173. The argument is augmented in Heyman’s “Addiction and Choice: Theory and New Data,” Frontiers of Psychiatry 4:31 (May 2013):1-5. doi: 10.3389/fpsyt.2013.00031.
- Heyman, Addiction: A Disorder of Choice , ibid., p. 173.
- __________, “Addiction and Choice: Theory and New Data,” op. cit.
- H. Hansen, “Faith-based Treatment for Addiction in Puerto Rico,” Journal of the American Medical Association 291:23 (June 16, 2004):2882.
- M. E. McCullough and B. L. B. Willoughby, “Religion, Self-Regulation, and Self-Control: Associations, Explanations, and Implications,” Psychological Bulletin 135 (2009):69-93. doi: 10.1037/a0014213.
- G. L. Xiong and P. M. Doraiswamy, “Longevity, Regeneration, and Optimal Health: Does Meditation Enhance Cognition and Brain Plasticity?” Annals of the New York Academy of Sciences 1172 (2009):63-69.
- R. J. Davidson et al., “Alterations in Brain and Immune Function Produced by Mindfulness Meditation,” Psychosomatic Medicine 65:4 (July 2003):564-570. doi: 10.1097/01.PSY.0000077505.67574.E3.
- McCullough and Willoughby, “Religion, Self-Regulation, and Self-Control,” op. cit., p. 84.
- See 1 Corinthians 10:13 (ESV). All Scripture texts in this article are quoted from the English Standard Version. Scripture quotations marked ESV are from The Holy Bible, English Standard Version, copyright © 2001 by Crossway Bibles, a division of Good News Publishers. Used by permission. All rights reserved.
- See also James 4:7 and Hebrews 2:18, as well as the implied promise of deliverance found in the prayer of our Lord in Matthew 6:13. Ephesians 6:11 to 18 also provides a list of spiritual disciplines that can serve as a protective shield against temptation.v




